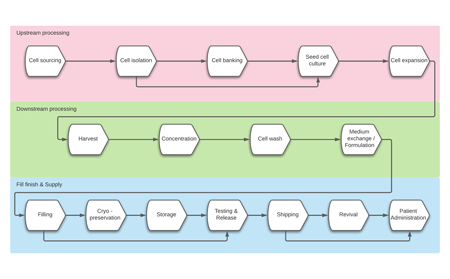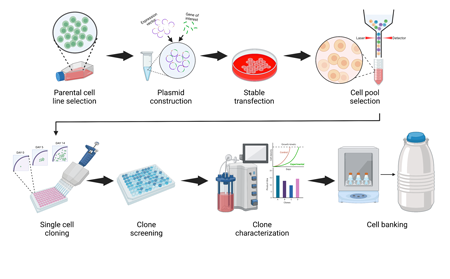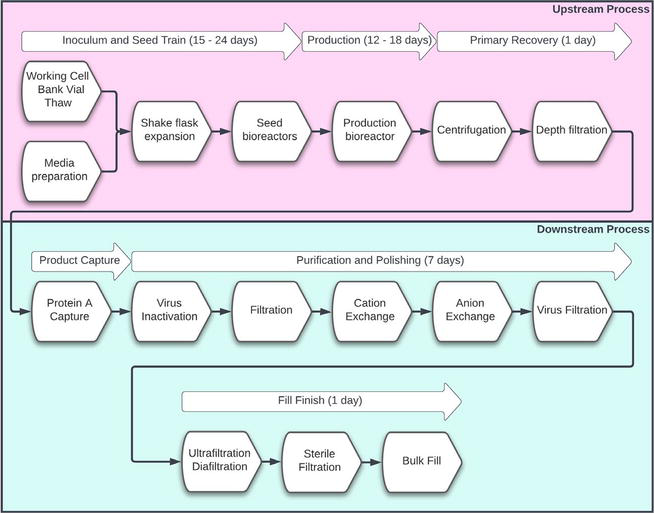
Featured Publication
Therapeutic antibodies dominate the biopharmaceutical market with continual innovations being made to provide novel and improved antibody treatment strategies. Speed to-market and cost-efficiency are of increasing importance due to the changing landscape of the biopharmaceutical industry. The increasing levels of competition from biosimilars, the increase in small volume products and political and social pressure to reduce the cost of treatments are some of the challenges currently being faced. Chinese hamster ovary (CHO) cells have been the workhorse in the production of therapeutic antibodies over the last 36 years due to the robust nature and high productivity of these cell lines. However, there are many biomanufacturing challenges remaining. The aim of this review is to examine the current biological, and engineering challenges facing the biomanufacturing of antibodies and to identify the mitigations and emerging technologies that can be employed to overcome them. Developments in cell line engineering, intensified processing, continuous manufacturing, automation and innovations in process analytical technologies and single use technology will be discussed with regard to their ability to improve the current performance of mAb production processes.